QClamp® KRAS Mutation Detection Tests
Improved Sensitivity for Single Gene Mutation Detection
The QClamp® KRAS Mutation Detection Test for plasma and Formalin-Fixed Paraffin-Embedded (FFPE) samples aids in the identification of cancer patients eligible for treatment and in monitoring response to therapy, which can lead to improved outcomes in cancer patients. The QClamp® KRAS Mutation Detection Test is an in vitro diagnostic real-time quantitative PCR assay for the detection of somatic mutations in and near codons 12 and 13 in Exon 2, codons 59 and 61 in Exon 3, and codons 117 and 146 in Exon 4 in the human KRAS gene, using purified DNA extracted from FFPE or plasma.
Powered by XNA technology, the QClamp® KRAS Mutation Detection Test has achieved a much higher analytical sensitivity compared to other commercial qPCR kits and other cancer gene mutation detection methods. QClamp® KRAS Mutation Detection Test is able to reliably detect 0.1% to 0.5% mutant DNA out of wild-type DNA for targeted mutations, providing lower detection limit compared to similar assays available in the market due to robust enrichment of mutant sequences while suppressing amplification of wild-type sequences.
Product Catalog
QClamp® KRAS Mutation Detection Test – CE Version: DC-10-3010
QClamp® KRAS Mutation Detection Test – Research-Use Version: DC-10-3010R
Pack Size: 30 Samples
Customized Kits Offering
Pick one or more existing NRAS target from the standard KRAS kit: Codons 12, 13, 59, 61, 117 and 146
Service Offering
We provide research service for QClamp® KRAS Mutation Detection Test.
GET A QUOTE NOW
Advantages of QClamp® KRAS Mutation Detection Test

ULTRA-SENSITIVE
Reliably detects 0.1% to 0.5% VAF mutant DNA out of wild-type DNA for targeted mutations

SAMPLE READY
Suitable for
plasma and FFPE samples

LOW INPUT DNA
Minimum 5ng input DNA per reaction. Less than 2 tubes of blood (10mL each) needed for cfDNA

COMPREHENSIVE COVERAGE
Covering all relevant somatic mutations in 6 codons of KRAS oncogene
FAST RESULTS
Less than 4 hours of
assay run time

GREAT VERSATILITY
Validated on the most common
qPCR machines with minimized variability
KRAS Mutation and Cancer
The Most Frequently Mutated Oncogene in Cancer
One of the common cancer gene mutations, Kirsten rat sarcoma viral oncogene (KRAS) mutations, are found in several cancers including pancreatic (80 to 90%), colorectal (25 to 60%), lung (25 to 60%) cancers, and are associated with poor prognosis. Mutations in KRAS may lead to abnormal growth
KRAS as an Important Biomarker for Targeted Therapy
KRAS is an effector molecule involved in the transduction of signals from the epidermal growth factor receptor (EGFR) to the nucleus. EGFR is overexpressed in metastatic colorectal cancer (
Recent Clinical Study
A recent clinical study found that about fifty
Supporting Data for QClamp® KRAS Mutation Detection Test
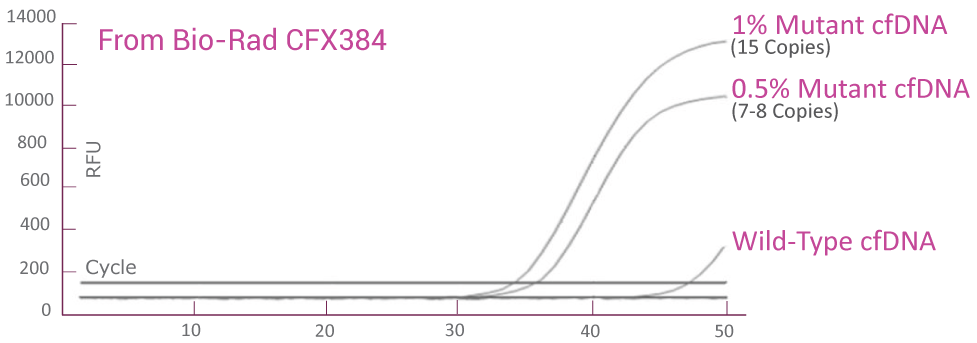
QClamp® KRAS Mutation Detection Test analytical sensitivity in plasma: Detects as low as 0.5% VAF mutant DNA in a 5ng input
cfDNA extracted from plasma of a colorectal cancer patient with known KRAS G12D mutation was diluted in 5ng/μl of wild-type cfDNA. VAF of the sample was verified by Next-Generation Sequencing (NGS). The KRAS c12 assay was able to detect as low as 7-8 KRAS G12D mutant gene copies (0.5% VAF) in the background of wild-type cfDNA.
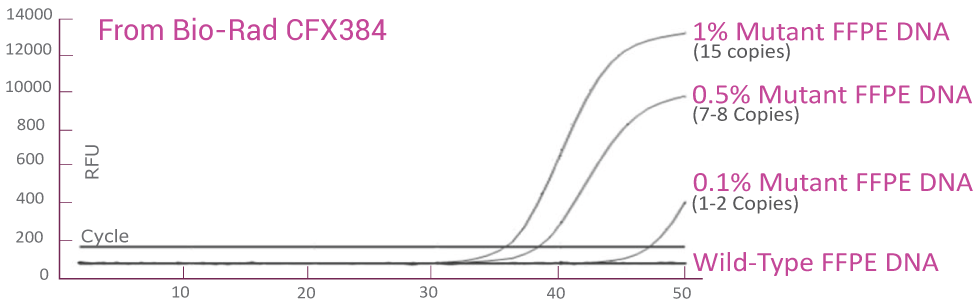
QClamp® KRAS Mutation Detection Test analytical sensitivity in FFPE: detects as low as 0.1% VAF mutant DNA in a 5ng input
DNA extracted from FFPE of a colorectal cancer patient with known KRAS G12D mutation was diluted in 5ng/μl of wild-type FFPE DNA. VAF of the sample was verified by NGS. The KRAS c12 assay was able to detect as low as 1.5 KRAS G12D mutant gene copies (0.1% VAF) in the background of wild-type FFPE DNA.
Streamlined Workflow for QClamp® Gene Mutation Detection Tests

Step 1: DNA Isolation & Quantification
Extract DNA from FFPE or plasma using a commercial DNA extraction kit and measure the concentration using fluorometric analysis

Step 2: set up qpcr
Mix the assay reagents, load into PCR plate, add controls and extracted DNA ~ 30-60 minutes
Step 3: Amplification parameters
Enter amplification parameters on
qPCR instrument, load PCR plate
and start the run ~ 2.5 hours
Step 4: Data analysis
Determine the presence or absence
of mutations according to the Cq
value cutoffs ~ 15 minutes
Resources
Catalog Number
Pack size: 10-sample
CE Catalog Number: DC-10-2010;
Research-use-only (RUO) catalog number: DC-10-2010R
Pack size: 30-sample
CE Catalog Number: DC-10-2020;
Research-use-only (RUO) catalog number: DC-10-2020R
Pack size: 60-sample
CE Catalog Number: DC-10-4020;
Research-use-only (RUO) catalog number: DC-10-4020R
Detected Codons
Codons 12, 13, 59, 61, 117 and 146
Sample Type
Plasma and FFPE
Input DNA
5-10ng/Reaction
Validated Instruments
Roche LightCycler® 480, Bio-Rad CFX384 and ABI QuantStudio 5
Detection Channel
FAM; HEX
Detection Chemistry
TaqMan
Turnaround Time
Less than 4 hours
Stability
Stable for 12 months at -25 ℃ to -15 ℃
Most frequent KRAS mutations detected by QClamp® KRAS Mutation Detection Test
| Exon | Amino Acid Change | Nucleotide change | Cosmic No. |
|---|---|---|---|
| 2 | G12>A | c.35G>C | 522 |
| G12>R | c.34G>C | 518 | |
| G12>D | c.35G>A | 521 | |
| G12>C | c.34G>T | 516 | |
| G12>S | c.34G>A | 517 | |
| G12>V | c.35G>T | 520 | |
| G12>D | c.38G>A | 532 | |
| G12>C | c.37G>T | 527 | |
| G12>R | c.37G>C | 529 | |
| 3 | A59>T | c.175G>A | 546 |
| Q61>H | c.183A>C & c.183A>T | 554/555 | |
| Q61>K | c.181C>A | 549 | |
| Q61>L | c.182A>T | 553 | |
| Q61>R | c.182A>G | 552 | |
| 4 | K117>E | c.349A>G | 1360831 |
| K117>N | c.351A>C & c.351A>T | 19940/28519 | |
| K117>R | 350A>G | ||
| A146>A | c.438A>G | 1360826 | |
| A146>G | c.437C>G | 1360829 | |
| A146>P | 436G>C | 19905 | |
| A146>T | 436G>A | 19404 | |
| A146>V | 437C>T>C | 19900 | |
Download
Ordering Information
For products that are in stock, DiaCarta will arrange shipment in 1-3 days. For products that are on backorder, DiaCarta will arrange shipment in 3-5 weeks.
Intended Use: QClamp® KRAS Mutation Detection Test is CE/IVD-certified. Outside the
Shipping Condition: QClamp® KRAS Mutation Detection Test will be shipped with dry ice. For domestic shipment, DiaCarta provides overnight delivery through FedEx Domestic Overnight Shipping Service. For international shipment, DiaCarta provides 3-7 days in transit through FedEx International Priority Shipping Service. Please contact DiaCarta if you prefer to use your shipping carrier.
